Table of Contents
Medications
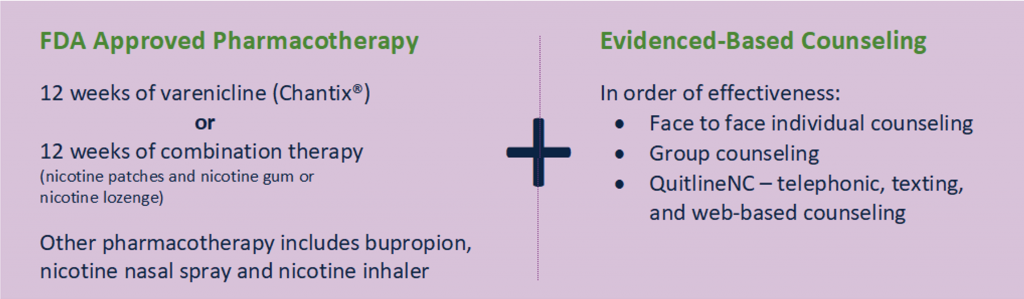
Evidence-based tobacco use treatment is medication assisted treatment.(1) The two medications that give clients the best chance of becoming tobacco free in the long term are Varenicline, also known as Chantix, and combination nicotine replacement therapy (usually a combination of nicotine patches + nicotine gum or lozenge).(2) This is why these two are highlighted in the North Carolina Tobacco Treatment Standard of Care for adults:
100% Tobacco free environments help people quit.
People with behavioral health conditions may require longer or more intensive treatment.
In the long run we know(1):
- With no medications or counseling less than 5% will quit.
- Counseling only and no medications, about 16% will quit.
- Combining intensive counseling and medications 32% will be successful on any one quit attempt.
- We want to give patients their best possible chance of this being the last time they have to try to quit smoking.
This is the standard of care for adults. More research is needed on a standard of care for adolescents who smoke, vape, or use tobacco. However, the American Academy of Pediatrics recommends that medical providers consider pharmacotherapy for youth who are moderately or severely addicted. Learn more from the American Academy of Pediatrics’ fact sheet: Supporting Youth Who Are Addicted to Nicotine: Advice for Pediatricians.
Starting medications two weeks before the quit day is helpful. It sets clients up for success by giving them time to learn how to use the medications, address any side effects and see if the dosage is fully treating their withdrawal. Someone who can cut down by half or more in the lead up to their quit day is making great progress.
We also know that varenicline or combination nicotine replacement therapy give the best odds a client will become tobacco free.(2) In 2017 Cahill et al. did a systemic overview and network metanalysis of research on tobacco use treatment medications, this review ultimately drew from hundreds of high-quality studies to make conclusions about the efficacy of tobacco use treatment medications.(2) They calculated the odds that a patient, given an exposure to a particular medication, would become tobacco free. Here are the odds ratios they calculated for the different medications(2):

Combination nicotine replacement therapy and varenicline were found to give the best odds compared to placebo for clients to become tobacco free.(2)
Check out this useful chart from the Mayo Clinic for evidence-based prescribing of tobacco use treatment medications. You can also review the resources available on improving prescribing in the Million Hearts Tobacco Cessation Change Package.
Nicotine Replacement Therapy
Nicotine replacement therapy (NRT) replaces the nicotine a person would normally get from tobacco (cigarettes, vaping/e-cigarettes, snuff/chewing tobacco) with something safe like the nicotine patch, gum or lozenge.(2) These products help people get past cravings while they learn how to live life tobacco free.(2)
NRT can include over-the-counter medications like the nicotine patch, gum or lozenge, and prescription medications like the nicotine nasal spray or nicotine inhaler.(2)
Combination NRT means combining a long-acting type of nicotine replacement, the nicotine patch, with a short-acting medication such as the gum, lozenge, inhaler, or nasal spray. The long-acting patch fights nicotine withdrawal symptoms all day, and the short acting is good for when the person experiences a trigger to use tobacco. For example, first thing in the morning or after a meal can be intense triggers for someone to smoke or vape, even with a patch on. The nicotine gum or lozenge can help to get through the cravings.
Clients often do not know that combination NRT is safe and effective or how to use NRTs. They may not realize that nicotine is the addictive part of cigarettes, vapes, and other tobacco products, but it is not the most dangerous part.(3) Tobacco products contain other chemicals that can cause cancer or make people sick.(3) NRTs do not contain these chemicals, they are safe and shown to work.(3)
Clients may have received too little NRT to treat their withdrawal symptoms or never used NRT in combination. This leads some folks think NRT will not help them or have concerns about using it. Use this patient education hand out: Nicotine Replacement Therapy: Using the Nicotine Patch, Gum, and Lozenge to educate yourself and clients about these important medications. It is also available in Spanish.
CDC also offers a website on how to use tobacco use treatment medications.
You can learn the most updated information about evidence-based prescribing of tobacco-use treatment medications including nicotine replacement therapies from a nationally accredited tobacco use treatment specialist program. North Carolina’s program is the Duke-UNC Tobacco Treatment Specialist Training Program. Other training resources can be found here.
To treat nicotine withdrawal effectively and safely onsite, organizations can offer the nicotine patch, gum, or lozenge to clients, visitors, and staff.
These products are over the counter, there is no need for a prescription, however there are many reasons an agency would need to prescribe for patients, and institute policies and procedures or a standing order to ensure Medicaid reimbursement and access to these medications onsite, learn more below.
Policies, Procedures, and Standing Orders for Nicotine Replacement Therapy
To treat nicotine withdrawal effectively and safely onsite, organizations can offer the nicotine patch, gum, or lozenge to clients, visitors, and staff. These products are over the counter, there is no need for a prescription. In certain settings clients may need a prescription, for example, they may need a prescription in order for insurance (including NC Medicaid) to pay for it. Some agencies will also need policies and procedures to allow clients who can self-administer NRT to do so.
If clients will not immediately see a medical provider who can prescribe and dispense the medication, In addition to policies and procedures, agencies may need to institute a standing order that allows staff to provide the right nicotine replacement therapy to the client.
Providing NRT onsite through policies, procedures, or a standing order makes sure clients have their withdrawal symptoms treated immediately, setting them up for success both with adhering to a tobacco-free policy and for being physically and mentally able to fully participate in services without having to fight withdrawal symptoms.
Work with your regional or local tobacco prevention and control staff to implement the right policies, procedures, and/or a standing order for medications at your organization.
Over-the-Counter Nicotine Replacement Therapy and Medicaid
Over-the-counter nicotine replacement therapies are covered by NC Medicaid. When prescribing or selecting nicotine patches, gum or lozenges for a patient/client who has Medicaid, be sure and pick a medication with the proper National Drug Code or NDC.
For a list of all Medicaid covered over-the-counter nicotine replacement therapies with NDC codes click here. Any nicotine replacement therapy not on this list, will not be covered by Medicaid. This may cause confusion and result in your patient not receiving their prescribed and covered tobacco pharmacotherapy. To ensure your patient’s/client’s pharmacotherapy is covered:
- Add in the Notes to Pharmacy section of your NRT prescriptions for Medicaid recipients: “Pharmacy – Please fill with a Medicaid-covered NDC. For a current list call NC Tracks at 1-800-688-6696.”
- If it is rejected, call the pharmacy and tell them to use one of the nicotine replacement options on the NDC list.
Varenicline (Chantix)
Varenicline is the generic name for the medication Chantix. Varenicline is a non-nicotine tobacco use treatment medication. When someone smokes, vapes, or uses tobacco, the nicotine in tobacco normally binds to certain receptors in the person’s brain in order to give feelings of pleasure, or at the very least relief from nicotine withdrawal.(2) Varenicline binds to these nicotine brain receptors and which makes the person have fewer cravings.(2) If the person smokes, vapes or uses tobacco on top of taking varenicline, the varenicline will block the nicotine in the tobacco products from attaching to those receptors. This blocks the pleasure that would normally come from smoking or vaping.(2) Clients will often say their cigarettes taste bad or are not pleasant as a result.
Varenicline is safe for people who have mental health or substance use disorder diagnoses and are stable.(4) A mental health condition is not a contraindication for using varenicline.(4) The FDA removed the black box warning for side effects on mood, behavior, or thinking from varenicline in 2016 after a massive 8000+ participant randomized controlled trial called the EAGLES trial.(4,5) The EAGLES trial, “compared the relative neuropsychiatric safety risk and efficacy of varenicline and bupropion with nicotine patch and placebo in smokers with and without psychiatric disorders.”(5)
The main finding from this trial was, “The study did not show a significant increase in neuropsychiatric adverse events attributable to varenicline or bupropion relative to nicotine patch or placebo.”(5) This was true for people with and without psychiatric disorders.(5) These outcomes were significant enough for FDA to determine that the risk of changes to thinking, behavior, and mood were much lower than previously thought and that the consequences for any of these side effects if they did occur were generally mild, leading them to remove the black box warning.(4)
You can learn the most updated information about evidence-based prescribing of tobacco-use treatment medications including varenicline from a nationally accredited tobacco use treatment specialist program. North Carolina’s program is the Duke-UNC Tobacco Treatment Specialist Training Program. Other training resources can be found here.
Accessing Medications for Uninsured People
QuitlineNC is a resource for nicotine patches, gum, and lozenges for people who are uninsured. Learn more about resources for prescription medications here.
References
- US Department of Health and Human Services. Treating Tobacco Use and Dependence: 2008 Update [Internet]. Rockville, MD: US Department of Health and Human Services; 2008 [cited 2021 Jun 15]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK63952/
- Cahill K, Stevens S, Perera R, Lancaster T. Pharmacological interventions for smoking cessation: An overview and network meta-analysis [Internet]. Vol. 2013, Cochrane Database of Systematic Reviews. John Wiley and Sons Ltd; 2013 [cited 2021 Jun 15]. Available from: https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD009329.pub2/full
- U.S. Department of Health and Human Services. Smoking Cessation: A Report of the Surgeon General. Atlanta, GA; 2020.
- U.S. Food & Drug Administration. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings [Internet]. 2016 [cited 2021 Jun 15]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-revises-description-mental-health-side-effects-stop-smoking
- Anthenelli RM, Benowitz NL, West R, St Aubin L, McRae T, Lawrence D, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): A double-blind, randomised, placebo-controlled clinical trial. The Lancet [Internet]. 2016 Jun 18 [cited 2021 Jun 15];387(10037):2507–20. Available from: http://www.thelancet.com/article/S0140673616302720/fulltext

